Medical Necessity Documentation:
Client Notes:
Patient Preparation:
Specimen Requirements:
Lesion swab collected in 3 mL viral culture media (VCM) or equivalent
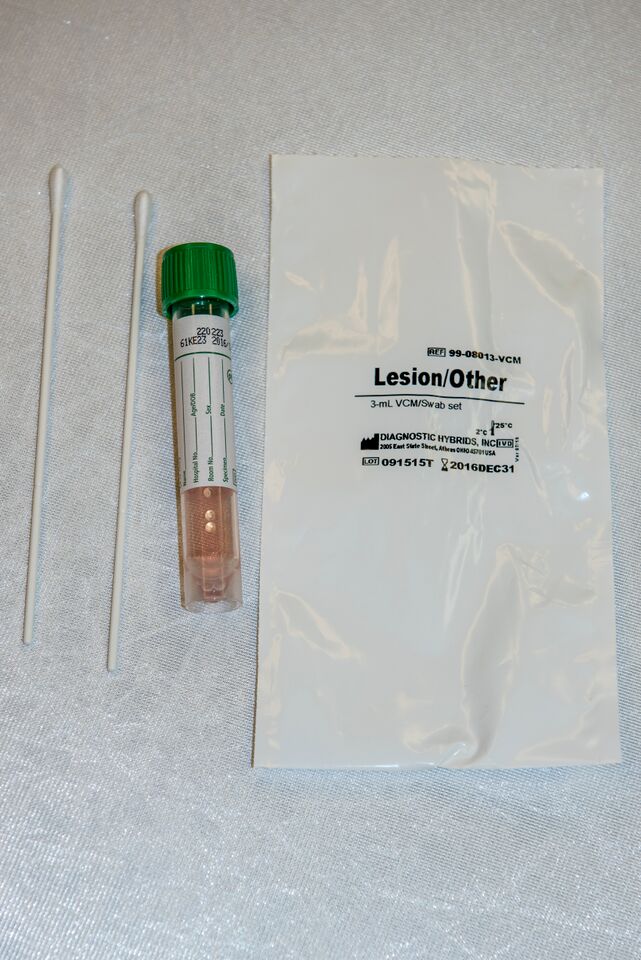
Collection Instructions:
Swab a pustule/lesion vigorously and place the swab into a viral culture media
Collection Instructons
Minimum Volume:
0.5 mL VCM media
Transport & Storage: Temperature/Stability:
Unacceptable Abmient
7 days Refrigerated
30 days Frozen (Transport)
Rejection Criteria:
Calcium alginate swabs • Cotton swabs • Wooden shaft swabs • Dry swabs (e.g. not submitted in VCM or equivalent)
Reference Range:
Orthopoxvirus DNA, QL PCR: Not detected
Monkeypox Virus DNA, QL PCR: Not detected
Critical Ranges:
Test Comments:
Methodology:
Real-Time Polymerase Chain Reaction (RT-PCR)
Clinical Significance:
The Monkeypox Virus DNA, Qualitative Real-time PCR test is intended for the qualitative detection of non-variola Orthopoxviruses and Monkeypox virus (West African clade) DNA using swabs from human pustular or vesicular rash specimensv>
Documentation:
No additional confirmatory testing is required at the CDC, therefore a duplicate swab from the same lesion is not needed. If clinically indicated, consider submitting additional swabs if multiple lesions with different stages are present. Multiple specimens collected on a single patient should be submitted separately and be accompanied by its own separate requisition and transported in its own sealed bag.
Assay Category FDA/EUA (Emergency Use Authorization)
Custom Panel:
No
PRODUCTION SCHEDULE
Turn Around Time:
2 to 3 days
Days Performed:
Sunday, Monday, Tuesday, Wednesday, Thursday, Friday, Saturday
Sites Performed:
Quest - Chantilly to San Juan Capistrano
PHL Test Code:
MONPV
EPIC Test Code:
LAB3242
Send Out Test Code:
12084
Alternate Test Names:
Mpox Virus DNA, Qualitative RT-PCR; MonkeyPox Virus DNA; Monkeypox Virus DNA, QL PCR; Monkeypox Virus DNA, Qualitative Real-Time PCR; Orthopoxvirus; Poxvirus
Included Tests:
Orthopoxvirus and Mpox Virus
CPT Coding:
87593 (x2)